Over the past decade, the psychedelics industry has been undergoing a second renaissance, evolving from a niche and legally questionable research area into a burgeoning field that promises to redefine mental health and wellness. As more companies, academic institutions, and non-profits explore the therapeutic potential of compounds like psilocybin, MDMA, and LSD, the role of intellectual property (IP) will become an ever more critical component for protecting innovation, securing investment, and ensuring sustainable growth – while at the same time stirring controversy over novelty and prior art, indigenous rights, and other hot button issues.
In the first part of this series, we aim to provide a very high-level overview of the current state of the psychedelics IP landscape and highlight the different approaches to IP taken by various stakeholders in the field. In subsequent articles, we will take deep dives on various IP strategies in the space and, importantly, explore the related valuation issues they bring up.
The Current State of the Psychedelics IP Landscape
The current state of the psychedelics field is characterized by a complex and sometimes dizzying intersection of science, regulation, public sentiment, health and wellness, history, and spirituality. With increasing interest in the potential of psychedelics across the globe, the role of IP in the field has emerged as a hot topic of discussion. Currently, the field faces several unique challenges:
- Regulatory Uncertainty
- Scientific Complexity
- Ethical Considerations
- Public Sentiment
Approaches to Psychedelic IP Development & Protection
Different organizations in the space are adopting varying approaches to IP. These approaches are often indicative of their underlying mission, business model (or lack thereof), and the specific compounds they are exploring.
Approach #1: Traditional IP Strategies
There is perhaps no industry more intimately familiar with, and dependent on, IP protection strategies than the pharmaceutical industry. Pharma companies file patents on novel compounds, methods of synthesis, formulations, and therapeutic protocols with the aim of creating a robust portfolio that not only protects the underlying science but is also critical to attracting and de-risking investments. Perhaps the best example of the traditional approach to IP protection in the psychedelics space resides with COMPASS Pathways.
Case Study: COMPASS Pathways
COMPASS Pathways is a leading company in the development of psilocybin therapies for treatment-resistant depression (TRD), post-traumatic stress disorder (PTSD), and anorexia nervosa. As detailed in the patent analytics figures below, the company has secured a portfolio of patents related to:
- Crystalline forms of psilocybin.
- Dosing regimens and administration methods.
- Specific therapeutic protocols that integrate psilocybin treatment with psychological therapy.
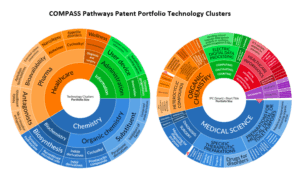
These patents have enabled COMPASS to build a strong competitive moat, attract significant venture capital funding, go public, and advance through advanced stages of clinical trials. By leveraging a traditional patent portfolio, COMPASS aims to demonstrate how classical IP strategies can be effective in navigating and succeeding in a space that is currently fraught with uncertainty, regulation, and competition.
Approach #2: Open Science and Collaborative IP Models
In contrast to the traditional “closed” model of IP protection pursued by the likes of COMPASS, other organizations are embracing open innovation approaches. These models emphasize collaboration, data sharing, and collective progress over proprietary exclusivity. By making R&D findings publicly available or via non-exclusive licensing, these organizations aim to accelerate scientific discovery and maximize broader public benefit.
Case Study: The Multidisciplinary Association for Psychedelic Studies (MAPS)
MAPS is a prominent non-profit organization that has for decades championed an open, collaborative model for advancing psychedelic research, particularly for MDMA-assisted therapies for post-traumatic stress disorder (PTSD). Instead of relying on the exclusionary nature of patents, MAPS:
- Prioritizes transparency in clinical trial data.
- Facilitates partnerships that encourage shared learning and resource pooling.
- Engages with the broader community of researchers and clinicians.
MAPS’ approach aims to enable a faster pace of research and has fostered a collaborative environment where innovations are rapidly disseminated, potentially lowering the cost of therapy development and broadening patient access.
Approach #3: Innovative Licensing & Partnership Models
Some players in the psychedelic space are exploring and leveraging innovative licensing and partnership models as an alternative, or a complement to, classical patent protection. These models are focused on using strategic collaborations and licensing agreements to maximize the reach and impact of their innovations. Their goals is to balance the benefits provided by exclusivity with those that can be derived from collaboration and broader access to therapies.
Case Study: Atai Life Sciences
Atai Life Science is an example of a player in the psychedelics space which is adopting this approach. Atai’s approach includes:
- Acquiring, licensing, and developing various psychedelic compounds and related technologies.
- Partnering with other biotech firms to share risks and accelerate clinical development.
Examples of such partnerships include Atai’s:
- Investment in COMPASS Pathways1
- Joint Venture in Entheogenix with Cyclica2
- Joint Venture in Invyxis with Dalriada Drug Discovery3
- License with Psilera4
- Joint Venture in TryptageniX with CB Therapeutics5
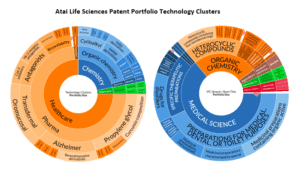
Through this partnership model, Atai has been able to diversify its risk and accelerate development timelines. This approach broadens the pipeline of therapeutic candidates and also creates a framework where innovation can be quickly adapted to meet varying regulatory and market conditions across different regions. The figure below shows the various categories of patented technology within Atai’s portfolio, not accounting for the broader of scope of Atai has access to through its various licenses and joint ventures.
Conclusion
The role of IP in psychedelics will no doubt continue to evolve. It’s clear from the examples above that there is no one-size-fits-all approach. Each model offers unique advantages and challenges, reflecting the multidimensional nature of the psychedelics world. It remains to be seen whether certain approaches will win out over others, or whether they will coexist in a world where a rising tide lifts all boats. No matter the model, it will be important for industry participants to take a thoughtful approach to create sustainable value.
As the landscape continues to mature, stakeholders who successfully navigate the journey will not only protect their innovations but also pave the way for more accessible and effective therapies that can potentially benefit millions around the globe. In the next part of our series, we will explore such strategies in further detail and analyze the related impacts on the valuations of the companies and their underlying IP assets.




